
A
Acetate Buffer (for Sepharose Coupling)
100 ml 500 ml 1 L
0.1M NaOAc (pH 4) 0.82 g 4.1 g 8.2 g
0.5 M NaCl 0.58 g 2.9 g 5.8 g
H20 to volume
Alternative Pathway
The following are mixed in 12 x 17 mm glass tubes:
25 µl of sample to be tested
10 µl of Mg/EGTA Stock-2
10 µl GVBo-3
30 µl rabbit RBC (5 x 108/ml GVBo)
1-Two-fold dilution up to 1:64 are typically used for human serum samples.
2-Mg/EGTA stock is 100 mM Mg2Cl2, 100 mM EGTA (this ensures no classical pathway
activation during assay)
3-GVBo=GVB++ or normal GVB (gelatin veronal buffer saline containing 0.1% gelatin, 0.15 mM CaCl2, 0.1 mM MgCl2) but with no Ca+ or Mg+
GVB is commercially available
Incubate @ 37ºC for 30 min w/ frequent mixing.
The reactions are stopped by adding 1 ml of ice-cold GVB0.
Unlysed cells are then pelleted by centrifugation and the extent of hemolysis is
quantified by spectrophotometry (420 nm).
Calculate % lysis for each dilution using the water blank as the 100% value.
Avidin column (SoftLink™-Promega)
Preadsorption (to block non-reversible sites)
1- Pour 1 ml (or 500 ml) bed vol into column.
2-Equilibrate with 1X PBS. 20 bed volumes.
3-Preabsorb non-reversible binding sites by washing with 5 mM biotin in PBS
(rate: 6 ml biotin/hr/ml of resin)
4-After 2 biotin volumes have passed through the column, stop flow for 15 min and
allow the biotin to bind.
5-Regenerate the column by passing 8 ml of 10% acetic acid followed by 20-30 m
l of PBS. Stop flow (with PBS covering the bed) and let sit for 30 min to allow avidin
to refold.
6. Use or store in 20% ethanol (@4ºC) or use.
PURIFICATION
1-Flush out 20% ethanol by washing with 10 ml PBS.
2-Load sample (for a 1 ml column, 4 mg protein can be added (6 ml/hr/ml of bed).
3-Wash with 5 ml PBS.
4-Elute with 10 mM biotin (0.024g + 600 µl DMSO to dissolve biotin
then add 9.4 ml PBS) in PBS.
5-Wash with 5 ml PBS.
6-Store in 20% ethanol
Working Solution: 1 ml stock in 39 ml saline, sterile filter, maintain
covered in foil and store on ice.
-Administer s.c. (0.16 µl/g).
-Approximately 350 µl/mse
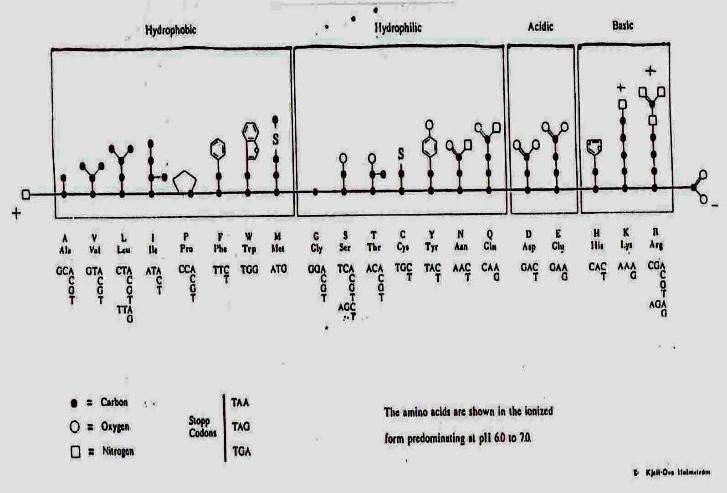
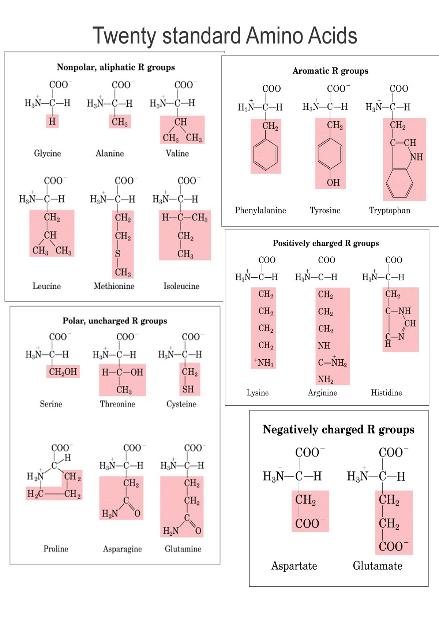
Amino Acids
Acetate Buffer (for Sepharose Coupling)
100 ml 500 ml 1 L
0.1M NaOAc (pH 4) 0.82 g 4.1 g 8.2 g
0.5 M NaCl 0.58 g 2.9 g 5.8 g
H20 to volume
Alternative Pathway
The following are mixed in 12 x 17 mm glass tubes:
25 µl of sample to be tested
10 µl of Mg/EGTA Stock-2
10 µl GVBo-3
30 µl rabbit RBC (5 x 108/ml GVBo)
1-Two-fold dilution up to 1:64 are typically used for human serum samples.
2-Mg/EGTA stock is 100 mM Mg2Cl2, 100 mM EGTA (this ensures no classical pathway
activation during assay)
3-GVBo=GVB++ or normal GVB (gelatin veronal buffer saline containing 0.1% gelatin, 0.15 mM CaCl2, 0.1 mM MgCl2) but with no Ca+ or Mg+
GVB is commercially available
Incubate @ 37ºC for 30 min w/ frequent mixing.
The reactions are stopped by adding 1 ml of ice-cold GVB0.
Unlysed cells are then pelleted by centrifugation and the extent of hemolysis is
quantified by spectrophotometry (420 nm).
Calculate % lysis for each dilution using the water blank as the 100% value.
Avidin column (SoftLink™-Promega)
Preadsorption (to block non-reversible sites)
1- Pour 1 ml (or 500 ml) bed vol into column.
2-Equilibrate with 1X PBS. 20 bed volumes.
3-Preabsorb non-reversible binding sites by washing with 5 mM biotin in PBS
(rate: 6 ml biotin/hr/ml of resin)
4-After 2 biotin volumes have passed through the column, stop flow for 15 min and
allow the biotin to bind.
5-Regenerate the column by passing 8 ml of 10% acetic acid followed by 20-30 m
l of PBS. Stop flow (with PBS covering the bed) and let sit for 30 min to allow avidin
to refold.
6. Use or store in 20% ethanol (@4ºC) or use.
PURIFICATION
1-Flush out 20% ethanol by washing with 10 ml PBS.
2-Load sample (for a 1 ml column, 4 mg protein can be added (6 ml/hr/ml of bed).
3-Wash with 5 ml PBS.
4-Elute with 10 mM biotin (0.024g + 600 µl DMSO to dissolve biotin
then add 9.4 ml PBS) in PBS.
5-Wash with 5 ml PBS.
6-Store in 20% ethanol
Working Solution: 1 ml stock in 39 ml saline, sterile filter, maintain
covered in foil and store on ice.
-Administer s.c. (0.16 µl/g).
-Approximately 350 µl/mse
Amino Acids
Blood Cell Counts
(WBCs)
-Normal levels
(WBCs)
-Normal levels

BCA standards (BCA Kit
Pierce)
-Make 1 mg/ml BSA Stock i.e., 100
µl 1% BSA + 900 µl PBS or 10 µl
10% BSA= 990 µl PBS
-10 µl Protein + 200 µl Working
Soln (Working Solution=40 µl
Reagent B + 2 ml Reagent A
-Incubate @ 37ºC/30 min
-Read @ 590 nm.
BCIP (see NBT or Western blot)
BCIP 0.075 g
DMF 5 ml
Store @ -20C in a glass vial wrapped in foil
(light sensitive)
BCIP 0.075 g
DMF 5 ml
Store @ -20C in a glass vial wrapped in foil
(light sensitive)
Bicarbonate buffer (10X, for
fluorescein labeling)
For 500 ml:
500 mM Na bicarbonate (NaHCO3)
(21 g)
QS to almost 500 ml, pH to 8.5, QS
to 500 ml, autoclave.
fluorescein labeling)
For 500 ml:
500 mM Na bicarbonate (NaHCO3)
(21 g)
QS to almost 500 ml, pH to 8.5, QS
to 500 ml, autoclave.
B
Biotinylation (using
Sulfo-NHS-LC-Biotin, Pierce)
-Dissolve 1 mg of NHS-LC-Biotin in 1 ml water
immediately prior to use.
-Add 74 µl of biotin soln to approximately 2 mg
protein in 1 ml PBS.
-Incubate for 2 h on ice or 30 min @ RT.
-Dialize into PBS with 3 changes of at least 3 L
each.
-Store protein @ 4ºC.
Biotinylation (using
Sulfo-NHS-LC-Biotin, Pierce)
-Dissolve 1 mg of NHS-LC-Biotin in 1 ml water
immediately prior to use.
-Add 74 µl of biotin soln to approximately 2 mg
protein in 1 ml PBS.
-Incubate for 2 h on ice or 30 min @ RT.
-Dialize into PBS with 3 changes of at least 3 L
each.
-Store protein @ 4ºC.
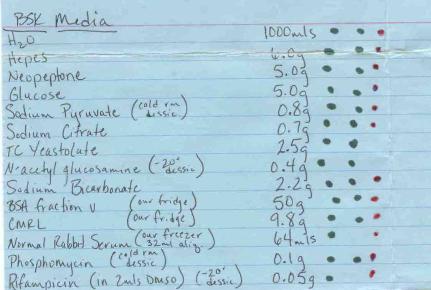
BSKII continued:
Stir for 1 h, pH to 7.5 w/ 10 M
NaOH, stir additional 20-30 min
and pH again.
Stir for 1 h, pH to 7.5 w/ 10 M
NaOH, stir additional 20-30 min
and pH again.
Borate buffer
100 ml 500 ml
0.1 M boric acid (pH 8.0) 0.62 g 3.1 g
0.5 M NaCl 0.58 g 2.9 g
QS to vol (with water).
100 ml 500 ml
0.1 M boric acid (pH 8.0) 0.62 g 3.1 g
0.5 M NaCl 0.58 g 2.9 g
QS to vol (with water).
BSKII
Binding Buffer/Buffer A (8X, for IDA column) also see IDA buffers.
100 ml 200 ml 500 ml
240 mM Imidazole 1.62 g 3.24 g 8.16 g
4M NaCl 23.4 g 46.8 g 116.9g
160 mM Tris HCL 16 ml 32 ml 80 ml
(1 M, pH 8.0)
pH to 7.9, filter and store at 4ºC.
100 ml 200 ml 500 ml
240 mM Imidazole 1.62 g 3.24 g 8.16 g
4M NaCl 23.4 g 46.8 g 116.9g
160 mM Tris HCL 16 ml 32 ml 80 ml
(1 M, pH 8.0)
pH to 7.9, filter and store at 4ºC.
Blue Juice (DNA loading buffer 1X)
1 g ficolll
1 ml glycerol
200 µl 50X TAE
0.01 g bromophenol blue
QS with dH20 to 10 ml
6X Blue Juice
40 ml 10 ml
0.25% Bromophenol blue 0.1 g .0125 g
0.25% xylene cyanol 0.1 g .0125 g
30% glycerol (in water) 12 ml (100% glycerol) 3 ml (100% glycerol)
QS to volume
Store at 4°C.
1 g ficolll
1 ml glycerol
200 µl 50X TAE
0.01 g bromophenol blue
QS with dH20 to 10 ml
6X Blue Juice
40 ml 10 ml
0.25% Bromophenol blue 0.1 g .0125 g
0.25% xylene cyanol 0.1 g .0125 g
30% glycerol (in water) 12 ml (100% glycerol) 3 ml (100% glycerol)
QS to volume
Store at 4°C.
Blood Agar (1 L)
30 g TSB + 13.5 g agar
QS to 1 L and autoclave (121°C for 15 min)
Let media cool down to about 55°C and add
5% Defibrinated sheep's blood. Mix well and
pour plates.
Purchase blood from Quad Five.
30 g TSB + 13.5 g agar
QS to 1 L and autoclave (121°C for 15 min)
Let media cool down to about 55°C and add
5% Defibrinated sheep's blood. Mix well and
pour plates.
Purchase blood from Quad Five.
Amonium persulfate
-for SDS-PAGE GELS
-10% solution in dH2O
Ampicillin (100 mg/ml)
-1 g in 10 ml dH2O; sterile filter using syringe filter.
-Store at -20ºC.
Annealing temperature
(#G+C)(4) + (#A+T)(2)=minimum annealing
temperature. Also use Melting
Temperature Calculator.
ATP (0.1 M)
Dissolve 60 mg ATP in 0.8 ml water.
Adjust pH to 7.0 with 0.1 N NaOH.
Adjust vol to 1 ml with water.
Store at -70ºC.
-for SDS-PAGE GELS
-10% solution in dH2O
Ampicillin (100 mg/ml)
-1 g in 10 ml dH2O; sterile filter using syringe filter.
-Store at -20ºC.
Annealing temperature
(#G+C)(4) + (#A+T)(2)=minimum annealing
temperature. Also use Melting
Temperature Calculator.
ATP (0.1 M)
Dissolve 60 mg ATP in 0.8 ml water.
Adjust pH to 7.0 with 0.1 N NaOH.
Adjust vol to 1 ml with water.
Store at -70ºC.
Avertin
1-Avertin (2,2,2 Tribromoethanol
(Aldrich T4, 840.2)
2-Tert-Amyl alcohol
-Add 15.5 ml Tert-Amyl alcohol to
the 25 g dark bottle of Avertin.
-Stir o/n @ 4ºC.
-Store @ 4ºC
1-Avertin (2,2,2 Tribromoethanol
(Aldrich T4, 840.2)
2-Tert-Amyl alcohol
-Add 15.5 ml Tert-Amyl alcohol to
the 25 g dark bottle of Avertin.
-Stir o/n @ 4ºC.
-Store @ 4ºC
Amonium acetate
7.5 M amonium Acetate (57.81 g);
QS to 100 ml, autoclave.
7.5 M amonium Acetate (57.81 g);
QS to 100 ml, autoclave.
API® 20 E Biocehmical Identification Procedure, adapted from bioMerieux, Inc.
Preparation of the Strip
1. Distribute approximately 5 mL of demineralized water into the wells of the incubation box tray.
2. Record the colony identification number on the elongated flap of the tray.
3. Remove the strip from its packaging and place in the tray.
Preparation of the Inoculum
1. Put 5 mL of 0.85% NaCl into a graduated Corning tube.
2. Using a pipette, remove three to five isolated colonies from a freshly grown (18-24 hours old) pure culture on MacConkey agar.
3. Emulsify and centrifuge to achieve a homogeneous bacterial suspension.
Inoculation of the Strip
1. With a pipette, suspend the bacterial suspension into the tubes of the strip. To avoid bubble formation, tilt the strip slightly forward and place the
pipette against the side of the cupule.
a. For the CIT, VP, and GEL tests, fill both the tube and the cupule.
b. For the remaining tests, fill only the tubes (not the cupules).
c. For the ADH, LDC, ODC, H2S, and URE tests, create anaerobiosis by overlaying with 2 to 3 drops of mineral oil.
2. Close the incubation box.
3. Incubate at 37°C for 18 to 24 hours.
Reading the Strip
1. If three or more of the 25 tests (GLU test + or -) are positive, record all reactions on the results sheet. Then, reveal tests which require the addition
of reagents:
a. TDA Test: add 1 drop of TDA reagent. A reddish brown color indicates a positive reaction.
b. VP test: add one drop of each VP1 and VP2 reagents. Wait at least 10 minutes. A pink or red color indicates a positive reaction. If a slightly pink
color appears after 10 minutes, the reaction should be considered negative.
c. IND Test: add 1 drop of JAMES reagent. A pink color developed in the whole cupule indicates a positive reaction. This test must be performed
last since this reaction releases gaseous products which interfere with the interpretation of other tests on the strip. The plastic incubation plate should not
be replaced after the addition of the reagent.
2. If the number of positive tests (including the GLU test) is less than 3:
a. Re-incubate the strip for another 24 hours without adding any reagents.
b. Reveal the tests requiring the addition of a reagent.
c. Perform supplementary tests, if necessary.
Preparation of the Strip
1. Distribute approximately 5 mL of demineralized water into the wells of the incubation box tray.
2. Record the colony identification number on the elongated flap of the tray.
3. Remove the strip from its packaging and place in the tray.
Preparation of the Inoculum
1. Put 5 mL of 0.85% NaCl into a graduated Corning tube.
2. Using a pipette, remove three to five isolated colonies from a freshly grown (18-24 hours old) pure culture on MacConkey agar.
3. Emulsify and centrifuge to achieve a homogeneous bacterial suspension.
Inoculation of the Strip
1. With a pipette, suspend the bacterial suspension into the tubes of the strip. To avoid bubble formation, tilt the strip slightly forward and place the
pipette against the side of the cupule.
a. For the CIT, VP, and GEL tests, fill both the tube and the cupule.
b. For the remaining tests, fill only the tubes (not the cupules).
c. For the ADH, LDC, ODC, H2S, and URE tests, create anaerobiosis by overlaying with 2 to 3 drops of mineral oil.
2. Close the incubation box.
3. Incubate at 37°C for 18 to 24 hours.
Reading the Strip
1. If three or more of the 25 tests (GLU test + or -) are positive, record all reactions on the results sheet. Then, reveal tests which require the addition
of reagents:
a. TDA Test: add 1 drop of TDA reagent. A reddish brown color indicates a positive reaction.
b. VP test: add one drop of each VP1 and VP2 reagents. Wait at least 10 minutes. A pink or red color indicates a positive reaction. If a slightly pink
color appears after 10 minutes, the reaction should be considered negative.
c. IND Test: add 1 drop of JAMES reagent. A pink color developed in the whole cupule indicates a positive reaction. This test must be performed
last since this reaction releases gaseous products which interfere with the interpretation of other tests on the strip. The plastic incubation plate should not
be replaced after the addition of the reagent.
2. If the number of positive tests (including the GLU test) is less than 3:
a. Re-incubate the strip for another 24 hours without adding any reagents.
b. Reveal the tests requiring the addition of a reagent.
c. Perform supplementary tests, if necessary.


